| Data Source | Description | Influencing Climatic Factors |
|---|---|---|
| Species Distribution and Range | Geographical presence and spread of mosquito species. | Temperature, Rainfall |
| Population Abundance | Number of individuals in a population. | Temperature, Humidity, Rainfall |
| Phenology (Seasonal Activity) | Timing of emergence, breeding, and dormancy. | Temperature, Seasonal Variation |
| Breeding and Oviposition Data | Egg-laying and larval habitat availability. | Rainfall, Humidity |
| Vector Competence and Disease Transmission | Ability to transmit pathogens. | Temperature, Humidity |
| Survival and Mortality Rates | Life expectancy and death rates. | Extreme Temperatures, Humidity |
| Dispersal and Flight Activity | Movement and spread of insects. | Wind Speed, Temperature, Humidity |
| Interaction with Natural Enemies | Predation and ecological interactions. | Temperature, Rainfall |
Understanding Malaria Data
Introduction
Malaria continues to pose a formidable challenge to global health, particularly in tropical and subtropical regions where transmission is sustained by favorable environmental conditions. Despite significant progress in reducing cases and deaths, the path to elimination requires not only robust surveillance but also advanced analytical approaches that can anticipate and respond to shifting transmission dynamics.
One of the most powerful tools in this effort is mathematical and computational modeling. By integrating malaria data with climatic variables such as temperature, rainfall, and humidity, models can capture the ecological drivers of transmission. These variables directly influence mosquito breeding, parasite development within vectors, and the seasonality of outbreaks. For example, rainfall patterns determine the availability of breeding sites, while temperature affects the speed of parasite maturation inside mosquitoes.
The benefits of modeling malaria with climatic inputs are substantial:
Predictive capacity: Models can forecast seasonal and geographic variations in transmission, enabling proactive interventions.
Resource optimization: By identifying high-risk periods and locations, health systems can allocate insecticide-treated nets, indoor spraying, and medicines more efficiently.
Policy guidance: Modeling provides evidence-based insights for national malaria control programs, supporting strategic decisions on elimination timelines.
Early warning systems: Climate-informed models can serve as surveillance tools, alerting authorities to potential outbreaks before they occur.
Ultimately, the integration of climatic variables into malaria modeling transforms raw data into actionable intelligence. This approach strengthens elimination efforts by shifting strategies from reactive responses to proactive, climate-sensitive planning. As malaria-endemic countries move closer to elimination goals, understanding and leveraging these models will be critical to sustaining progress and preventing resurgence.
Data types used to describe malaria transmission
Understanding malaria transmission requires the integration of diverse data types that capture the complexity of the disease’s dynamics. Epidemiological data which includes case counts, prevalence, incidence rates, and demographic information, which help quantify the burden of malaria in populations. Entomological data that focuses on mosquito vector characteristics such as species distribution, biting rates, and sporozoite infection rates, which are critical for assessing transmission potential. Environmental and climatic data encompasses temperature, rainfall, humidity, and land use patterns, as these factors influence mosquito breeding and parasite development. And finally, genetic and molecular data provides insights into parasite and vector genetics, drug resistance markers, and transmission-blocking interventions.
Combining these data types enables researchers to model transmission patterns, identify hotspots, and design targeted interventions for malaria control and elimination.
2.1 Summary of Malaria Entomological Data Types
A brief explanation of the main entomological data collected for progrsmme planning. The reason for collection of each data type is visited briefly and is clarified using an example of what the data type outputs.
| Data Type | Purpose | Examples |
|---|---|---|
| Species Distribution | Identify malaria vector presence and range | Mapping Anopheles species in different regions |
| Population Abundance | Estimate vector density for transmission risk | Adult mosquito counts per trap/night |
| Breeding & Oviposition | Locate larval habitats and predict population | Water body surveys; larval density counts |
| Phenology (Seasonality) | Understand seasonal activity and peak biting | Monthly biting rate trends |
| Vector Competence | Assess ability to transmit Plasmodium | Sporozoite infection rates in mosquitoes |
| Survival & Mortality | Determine longevity for parasite development | Adult survival under different temperatures |
| Dispersal & Flight | Track movement between habitats and dwellings | Mark-release-recapture studies |
| Human-Vector Contact | Measure exposure risk to humans | Human landing catches; indoor vs outdoor biting |
| Insecticide Resistance | Monitor effectiveness of control interventions | WHO susceptibility tests; genetic resistance markers |
Collecting and analyzing entomological data is fundamental for malaria control programs. These data types help in identifying transmission hotspots, monitoring vector behavior, and evaluating intervention effectiveness, ultimately contributing to reducing malaria burden globally.
2.2 Climatic Variables that influence entomological Data
| Climatic Variable | Influenced Entomological Data | Examples / Notes |
|---|---|---|
| Temperature | Species distribution; Population abundance; Phenology (seasonal activity); Vector competence | Warmer temperatures expand mosquito ranges; accelerate pathogen replication inside vectors; alter timing of insect emergence |
| Rainfall | Breeding/oviposition sites; Population abundance; Survival rates | Heavy rains create mosquito larval habitats; drought reduces breeding success; flooding can wash away larvae |
| Humidity | Survival/mortality rates; Activity levels; Vector competence | High humidity reduces desiccation risk; low humidity increases mortality in small-bodied insects |
| Wind | Dispersal and migration; Flight activity | Strong winds aid long-distance dispersal (e.g., locust swarms); storms can transport insects across regions |
| Seasonality | Phenology; Breeding cycles; Disease transmission patterns | Seasonal changes dictate insect dormancy (diapause), peak breeding times, and vector-borne disease outbreaks |
| Extreme Events (heatwaves, droughts, storms) | Mortality; Distribution shifts; Breeding success | Heatwaves cause mass die-offs; drought limits breeding; storms redistribute insect populations |
3. Data Correction of Malaria Entomology Data
Accurate data correction is essential for ensuring reliability in vector surveillance, intervention evaluation, and epidemiological modeling. The information presented is based on best practices and WHO guidelines.
3.1 Forms of Data Correction in Malaria Entomology
Malaria entomology data often requires multiple layers of correction to address errors from field collection, laboratory analysis, and data entry. These corrections include data cleaning, sampling effort adjustments, spatial and temporal alignment, laboratory verification, environmental integration, and indicator calculation checks.
3.1.1 Correction Checklist
The following checklist provides a structured approach to ensure data integrity:
| Category | Correction steps |
|---|---|
| Data Cleaning | Remove duplicates; Standardize species names; Validate dates; Check ranges; Handle missing values |
| Sampling Effort | Normalize counts by trap-nights; Adjust HLC for exposure time; Account for seasonality |
| Spatial & Temporal Alignment | Verify GPS coordinates; Align data to consistent time intervals |
| Laboratory Verification | Validate PCR/ELISA results; Reconcile species identification discrepancies |
| Environmental & Covariate Integration | Correct weather data; Incorporate intervention coverage |
| Indicator Calculation | Confirm formulas for EIR, HBR, Parity Rate; Ensure consistency across datasets |
Implementing these data correction steps ensures high-quality malaria entomology datasets, enabling accurate calculation of key indicators such as Entomological Inoculation Rate (EIR) and Human Biting Rate (HBR). This process supports evidence-based decision-making for malaria control and elimination programs.
3.2 Abbott’s Formula and Its Application
Measuring mortality among malaria vectors is often the endpoint of most vector assays and it is important that negative controls are done as part of the assay. If mortality is less than 5% then the results can be used without correction. If mortality is >5 ≤ 20%, the results need to be corrected to take into account mortality in the negative control. Abbott’s formula is a widely used correction method in entomology and bioassays. It helps researchers accurately assess the effectiveness of insecticides or treatments by accounting for natural mortality observed in control groups. This section summarizes the concept, mathematical expression, applications, and importance of Abbott’s formula, particularly in malaria vector studies.
3.2.1 Definition of Abbott’s Formula
Abbott’s formula was introduced by Walter S. Abbott in 1925. It is designed to correct observed mortality in treated insect populations by considering the mortality that occurs naturally in untreated controls. This ensures that the calculated effectiveness of a treatment is not overestimated.
Mathematical Expression
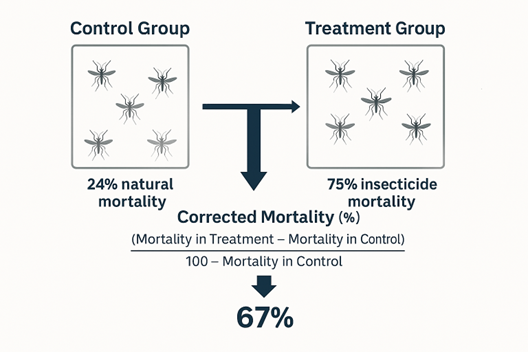
\[ \text{Corrected Mortality (\%)} = \frac{\text{Mortality in Treatment} - \text{Mortality in Control}}{100 - \text{Mortality in Control}} \times 100 \]
Use Cases
Abbott’s formula is commonly applied in the following scenarios:
- Insecticide susceptibility tests (e.g., WHO cone bioassays, CDC bottle bioassays)
- Malaria vector control studies, including IRS (Indoor Residual Spraying) and LLIN (Long-Lasting Insecticidal Nets) evaluations
- Resistance monitoring programs for mosquito populations
- Any bioassay where natural mortality in controls could bias results if not corrected
Importance
Abbott’s formula is crucial for ensuring accurate interpretation of bioassay results. Without this correction, high control mortality could lead to misleading conclusions about treatment efficacy. The formula standardizes results across experiments and is recommended by WHO guidelines for vector control evaluations.
Researchers conducting malaria vector studies should consistently apply Abbott’s formula when control mortality is observed. This practice enhances data reliability and supports informed decision-making in vector control programs.
3.2.2 Worked Example
Suppose mortality in the treatment group is \(75%\), and mortality in the control group is \(24%\). Using Abbott’s formula:
\[ \text{Corrected Mortality (\%)} = \frac{75 - 24}{100 - 24} \times 100 = \frac{51}{76} \times 100 \approx 67\% \]
This means the insecticide caused \(67%\) mortality after adjusting for natural deaths.
3.2.2.1 Diagram of Correction Process
4. Entomological Data Sources
| Source name | Description | Access link |
|---|---|---|
| WHO Global Malaria Programme | Official WHO tools for entomology and vector control data collection and visualization. | https://www.who.int/teams/global-malaria-programme/prevention/vector-control/dhis-data-collection-and-collation-tools |
| Global Mosquito Observations Dashboard (GMOD) | Citizen science platform for global mosquito observations. | https://sdgstoday.org/dataset/global-mosquito-observations-dashboard |
| MalariaGEN Mosquito Data Tools | Genomic data and analysis tools for mosquito populations. | https://www.malariagen.net/resources/mosquito-data-tools/ |
| MosquitoDB (Ifakara Health Institute) | Open-source system for managing mosquito field and lab data. | https://github.com/IHI-Data-science/mdb |
| VectorByte – Vector Data Ecosystem | Global repository for vector occurrence, infection, and geospatial data. | https://www.vectorbyte.org/vecdataecosystem |
| Smithsonian Entomology Collections | Digitized specimen data from one of the largest entomology collections. | https://collections.nmnh.si.edu/search/ento/ |
| Global Biodiversity Information Facility (GBIF) | Worldwide species occurrence records, including mosquitoes. | https://www.gbif.org |
| EMBL-EBI Biodiversity Portal | Biomolecular and genomic data for biodiversity and climate research. | https://www.embl.org/news/science-technology/embl-ebis-open-data-resources-for-biodiversity-and-climate-research/ |